B) False
Correct Answer

verified
Correct Answer
verified
True/False
A Galvanic cell is an electrochemical cell in which a spontaneous chemical reaction may be used to generate an electrical current.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rusting witnessed in the sheet metal of a '65 Impala is an example of:
A) uniform corrosion.
B) galvanic corrosion.
C) electrolysis.
D) compounding corrosion.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The standard hydrogen electrode (SHE) involves gaseous HCl at 1 atm being bubbled into water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the decomposition of hydrogen peroxide, which atom is being reduced? H2O2( aq ) → H2( g ) + O2( g )
A) H
B) O
C) both
D) neither
F) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
What is the E0 value for the Galvanic cell formed from these two half-reactions?
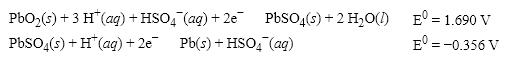
A) +1.334 V
B) − 2.046 V
C) +2.046 V
D) +2.758 V
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the formation of iron (II) oxide, which species is being reduced? Fe( s ) + O2( g ) → FeO( s )
A) FeO
B) Fe2O3
C) Fe
D) O2
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
True/False
Reduction occurs at the anode of a Galvanic cell.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given a Galvanic cell: 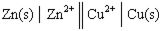 , the right-hand side of this notation represents the:
, the right-hand side of this notation represents the:
A) spontaneous half of the reaction.
B) oxidation 1/2 reaction.
C) anode of the cell.
D) reduction 1/2 reaction.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Secondary cells are designed to be rechargeable.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of Mn in MnO4 − ?
A) 1+
B) 2+
C) 7+
D) 8+
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the E0 value for the Galvanic cell formed from these two half-reactions?

A) − 2.31 V
B) +4.33 V
C) − 1.32 V
D) +2.00 V
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of silver are deposited at a platinum cathode in the electrolysis of AgNO3 (aq) by 5.30 amps of electric current in 4.0 hours?
A) 85.3 g
B) 42.6 g
C) 121 g
D) 188 g
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Galvanic corrosion may occur only when two different metals are in contact with one another.
B) False
Correct Answer

verified
True
Correct Answer
verified
Multiple Choice
In a redox reaction:
A) there is a transfer of protons from one species to another.
B) electrons are created.
C) electrons are transferred between two species.
D) the reaction always moves in a reverse direction.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the formation of iron (II) oxide, which species is being reduced? Fe( s ) + O2( g ) → FeO( s )
A) FeO
B) Fe2O3
C) Fe
D) O2
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following electrochemical reaction in acid: MnO4 − ( aq ) + Zr( s ) ↔ Mn2+( aq ) + Zr2+( aq )
A) 2 MnO4 − ( aq ) + 16H+( aq ) + 5 Zr( s ) → 2 Mn2+( aq ) + 5 Zr2+( aq ) + 8 H2O( l )
B) 2 MnO4 − ( aq ) + 5 Zr( s ) → 2 Mn2+( aq ) + 5 Zr2+( aq )
C) MnO4 − ( aq ) + Zr( s ) → Mn2+( aq ) + Zr2+( aq ) + 5 e −
D) 2 MnO4 − ( aq ) + 8H+( aq ) + 5 Zr( s ) → 2 Mn2+( aq ) + 5 Zr2+( aq ) + 8 H2O( l )
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Species with positive standard reduction potentials are excellent candidates for reduction reactions.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the provided standard reduction potentials, which of the following cells is an example of a Galvanic cell?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which phrase best describes the half-reaction as written? Na+ + e − → Na
A) oxidation 1/2 reaction
B) reduction 1/2 reaction
C) group 2 ionization
D) two electron transfer
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 40
Related Exams